It was a warm day for November, 51°F, and ants come out of dormancy when it gets warm. They need sustained warmth to become active again, and a few hours weren’t really enough, but it was a glorious day so why not go poking around looking for ants? I was taking an ant class, and newly armed with ant ID knowledge I wanted to test it out. But live ants will have to wait, I did not see one–all my ants must have been sound asleep. The leaf litter was alive, though, with moving, jumping, and gleaming things.
I walked through dried leaves on the trail, listening in delight as they rustled and crunched. Looking up, most trees had lost their leaves. There were the golden brown leaves of beech and oak, which persist into winter, and the occasional lemon-yellow foliage of a maple that did not want to give up. But I wanted to see the little stuff, so I dropped to my knees. A fuzzy white cocoon, 4mm in length, was lying in the curve of a crumbled bit of no longer identifiable leaf. Its form was perfectly elliptical, it gleamed clean and bright. Moth? I wish I knew, but I wish I knew a lot of things. A nearby leaf had been artfully skeletonized. The shape and direction of veins suggested a Maianthemum, a Canada mayflower. So much to see, and I had not even started to look.

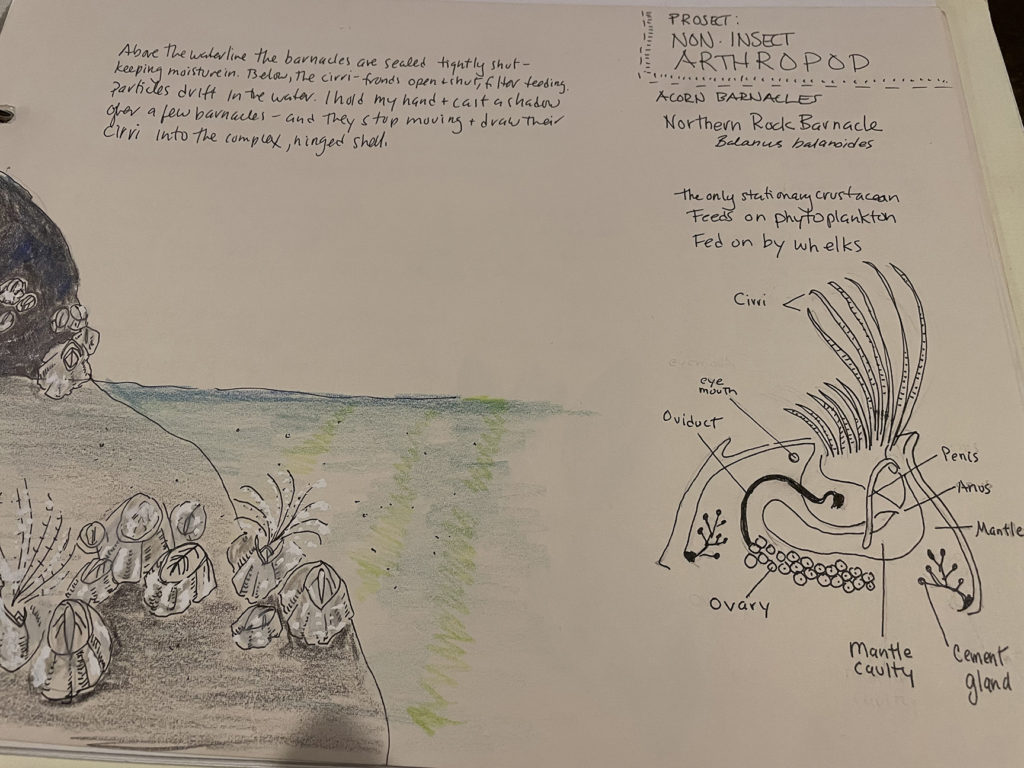
Sweeping the dry leaves off the soil I saw one or two quick-moving creatures run for cover. Too fast for me, I noted their glossy black beetle-like forms and let them go. Instead I leaned down to the exposed earth and inhaled. I never had a favorite rock star, but suddenly understood “swoon-worthy.” The rich, moist, peaty scent rocked me back on my heels with closed eyes. Finally I opened them and put my nose to the ground and my hand lens to my eye. Light was reflecting off a small coil. I rolled it onto my hand, and watched the coil unwind into a millipede and wander my palm. It eventually climbed down the side of my hand and back home. These common arthropods often curl up to avoid danger. This one uncurled instead, letting me see its coordinated prancing legs, not a thousand as its name might imply, but up to two hundred, and more than I could count. Along this gentle creature’s sides are two rows of ozopores, the openings to glands which produce a defensive odor consisting of cyanide, hydrochloric acid, and other irritating chemicals. It is enough to keep away a hungry toad, but not harmful to humans. Another very cool millipede fact is that they, (specifically an ancestor called Pneumodesmus newmani,) have been around 420 million years, and while there is a bit of argument, they were probably the very first animal to step foot, well, hundreds of feet, onto land. I sent a photo to an expert at the Field Museum in Chicago, and they ID’d this as the blunt-tailed snake millipede, Cylindroiulus punctatus.

Not far away, under the litter in some loose decomposing wood, were a half dozen white spherical eggs, and several coiled snails. There were two very distinct snail types, both about 1.5mm long. One was flattened and a golden yellow color, the other was shaped like a whelk, with a pointy top and brown and maroon coloring. Having never tried to ID snails I did not realize it would be so much more difficult than the millipede. Will someone please create a guide to terrestrial gastropods of Maine? Gastropod is from the Greek words gaster and podos, belly and foot. Several months ago in a New York second-hand bookstore I felt compelled to pick up a 1939 copy of the illustrated Fieldbook of Illinois Land Snails by Frank Collins Baker. Even if it is not terribly useful for Maine snails, the guide, with its clear introduction to gastropod morphology and life cycles and meticulous line drawings, is now at the top of my teetering bedside book pile. The UMaine fact sheet on slugs and snails is primarily about how to eradicate them, not at all what I was looking for, but at least informs that we have about 90 species here. I am now finding references to a guide without images, and will hunt that down after I am done hunting through leaf litter. iNat suggests family Pristilomatidae, but there are no known observations in Maine. The snail hunt goes on.
I brought the yellowish one home for a visit, and placed it under my microscope. The snail remained stationary and I was examining its pale, slightly translucent shell and shadowy occupant when an even smaller animal ambled along. This was a soft-bodied mite, and I had seen at least a half dozen out in the litter, some red, some brown, some black. In the subclass Acari, the one with the snail was tentatively keyed to the genus Oribotritia. I had watched one in the field as it slowly excreted dark frass. I will now recognize this mite scat, which came out with undulations like soft serve ice cream, whenever I encounter it.

There were so many fragments of life in the soil. An entire discarded larval case, and many smaller bits and pieces of exuvia. A rather large (comparatively speaking, it was maybe a mammoth 8mm) chunk of compacted soil began to tremble and then before my astonished eyes seemed to grow legs, shake off soil, and reveal itself as a leafhopper larva, Agallia sp. I brought it home, photographed it, then took it back before looking it up. I can confirm that it hopped, it did several times, right out of the petri dish in fact, but it seemed to walk in a straight line, and the literature says they move sideways.

You probably have leaf litter near you, so crouch down and take a look at all the life. This life feeds birds, and eventually feeds us. If you do not want leaf litter in your yard, drag it into a compost pile. The thousands of living creatures may get a bit tumbled, but will go on. Burning it will just create a massive smoke cloud, and certain death to thousands of harmless beings. So much life is overwintering in the leaf litter, patiently awaiting the return of spring, which will also wake up my sleepy ants. Then I can pick up a groggily awakening ant, and finally sharpen my ant ID skills.